
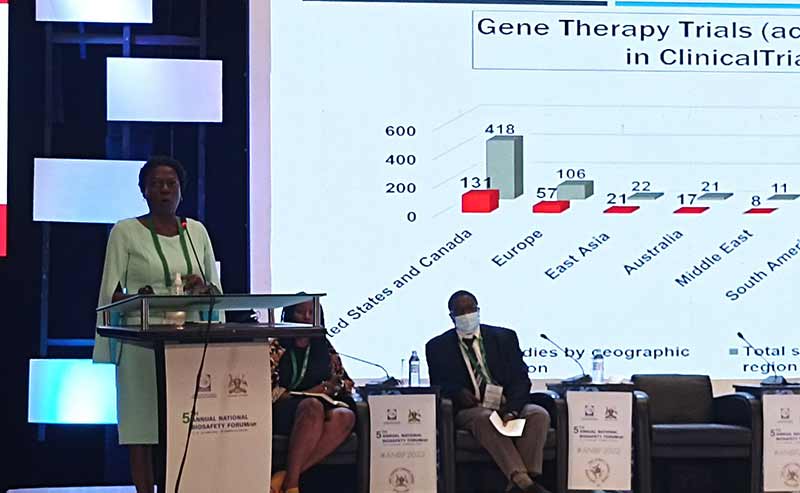
JCRC’s Dr. Cissy Kityo presenting about the opportunities of Gene Therapy for defeating diseases such as HIV, Sickle Cell and COVID
Gene therapy or the science of using genetic engineering to produce new drugs and vaccines for the treatment and control of complex health challenges in Uganda, has triggered a change in the mindset of leaders from one of mistrust to one of embracing the modern technology as a tool to address human diseases as well as agricultural-based challenges.
The sense of hope comes against the background of successes recorded in the rapid development of vaccines developed for the control of the COVID-19 pandemic using genetic engineering.
During last week’s 5th Annual National Biosafety Forum 2022 held at the Kampala Serena hotel, experts and participants engaged in a lively discussion about the potential of gene therapy in Uganda.
Experts from the Entebbe-based Uganda Virus Research Institute (UVRI) led by Prof. Pontiano Kaleebu revealed that they are working with the London School of Health and Tropical Medicine in the United Kingdom to develop COVID-19 vaccines using gene editing technology.
Dr. Sheila Balinda from UVRI informed the gathering of more than 200 participants that the COVID-19 vaccine development is undergoing tests in mice with the hope that clinical trials (trails involving humans), will be undertaken soon.
She further explained that UVRI is also working with international partners to develop a vaccine is designed to specifically target the unique HIV subtype that is found in Sub Saharan Africa.
During the same session, Dr. Cissy Kityo Mutuluza, the Executive Director of the Joint Clinical Research Centre (JCRC) based at Lubowa along Entebbe road, strongly argued that gene therapy offers the best chance of overcoming some of the most debilitating diseases in Uganda such as Sickle Cell and HIV/AIDS.
Dr. Kityo said that gene therapy would not only enable countries to reduce the disease burden that claims lots of lives and resources, it promises to generate money for the country using modern, one-time treatment for some of the most problematic diseases in Uganda.
Already, Dr. Kityo pointed out, JCRC has partnered with over a dozen institutions worldwide including The Bill and Melinda Gates Foundation and the National Institutes of Health (NIH), to accelerate access to cures developed through gene therapy.
The partnership is targeting access to treatment of three major debilitating diseases; HIV, B-Thalassemia and Sickle cell disease that have until now defied conventional chemical-based therapies.
According to Dr. Kityo, first clinical trails for developing cures for Sickle Cell and B-Thalassemia diseases being developed under the Global Gene Therapy Initiative (GGTI) partnership are expected to start in 2024.
Although Uganda has guidelines for scientific studies in restricted setting for products of genetic engineering, Kityo echoed the cry of most of her peers in the field of human and agricultural fields to introduce regulations that will allow not frustrate development of products through gene therapy.
She argued that absence of enabling laws and regulations mean that Uganda risks eliminating herself from actively participating in the emerging multi-billion dollar bio-economy.
The continuing high prevalence of HIV in Uganda (estimated at 1.4m persons with HIV) and other Sub Sahara African countries, coupled with donor fatigue, Kityo argued present an opportunity for Uganda to intensify the hunt for a cure.
“Uganda could become a regional centre of excellence, get intellectual property and generate money from products of gene therapy,” said Kityo before adding that her team has help meetings with President Yoweri Museveni to integrate gene therapy into the Genetic Engineering and Regulatory Act (GERA)
The Minister of Science, Technology and Innovation Dr. Monica Musenero also confirmed in her remarks while officiating at the opening of the Biosafety Forum that there are plans to review the to integrate gene therapy for use in human treatment.
Research to develop products of Genetic Engineering started in the agricultural sector when the Ugandan government in 2003, opened the first Biotechnology Laboratory at Kawanda, on the outskirts of Kampala. However over the years, rollout of the technology to smallholder farmers, has suffered from turbulence arising from negative campaigns in Europe and America aimed at frustrating its uptake in Africa on grounds that it would undermine food security by expanding the power of multinational seed companies.
Currently there is no law to regulate the commercialization of products of genetic engineering, be they for human health or for agricultural purposes, despite close to 20 years of successful research efforts especially in the agricultural field.
During her remarks at the 5th ANBF2022, Musenero suggested that the law on genetic engineering was ill-timed way back, and that now is the right time to debate such regulations. Dr. Musenero is a veterinary doctor and she has recently been involved with efforts to develop and GM-derived anti-tick drug.
Experts from the agricultural sector welcomed the apparent positive shift in the mindset of the leaders, with some noting that the era of genome editing presents even more opportunities for tailoring solutions to challenges.
Clet Wandui Masiga, the Executive Director of the Tropical Institute for Development Alternatives (TRIDI) said: “We are grateful that the new leadership has taken on what had been initiated. Of course we have lots of developments that happen every day. Using genome editing, we’re thinking about using tailor-made traits that we may require changing a few things in the law. But on the whole, the basic principles were well grounded in the previous draft. What we need is to enhance one or two things.”
Masiga added: “The Minister talks about the need to develop home growth products using genetic engineering. We were doing this before, although we were almost at the stage where many scientists were at the stage of rethinking where else to practice biotechnology (after the government backtracked on its plans to champion the technology in the region.)”
Masiga argues that following a period of uncertainty in the scientific community that was engaged in agricultural biotechnology development, the government needs to re-assure the scientists by among other things, removing some of the stringent provisions in the proposed law. One of the most controversial provisions is one that seeks to impose criminal sanctions on individual scientists and companies in case of adverse consequences resulting from the application of biotechnology – what is technically known as strict liability.
Masiga confessed that the scientific community had become frustrated with the government’s position on the technology.
He said: “I really have to be honest that we had chosen to take a back seat and rethink where we can best use our skills. We really need a lot of commitment to further invest in this field. Otherwise we shall look for where we can safely practice biotechnology and the government will have no choice but to import products of biotechnology from those other countries.”
Masiga said: “We are only happy that COVID-19 came and opened the eyes of our leaders that genetic engineering is the right technology to use.
At one of the sessions during the forum, Prof. David Kabaasa, the Principal College of Makerere University College of Veterinary, Animal Resources and Biosecurity strongly argued that formalizing science using a targets and a value-chain approach is critical in economic transformation.














Henry Lutaaya
Leave a Comment
Your email address will not be published.